Liquidia Corp. ($LQDA)
A Transformative Opportunity in Drug Commercialization - Yutrepia™
Welcome again to Strategic Alpha! Over the past few months, I've been closely observing Liquidia Corporation (LQDA), a promising player in the biopharmaceutical. So today we deep dive into the company's financials, release of a new drug, market potential, and strategic positioning.
Buckle up… as we explore why Liquidia is capturing the attention of investors and what the future may hold for this company!
Table of Contents
Introduction
Company and Market Overview
Recent News and Developments
Detailed Financial Analysis
Valuation Analysis
1. Introduction
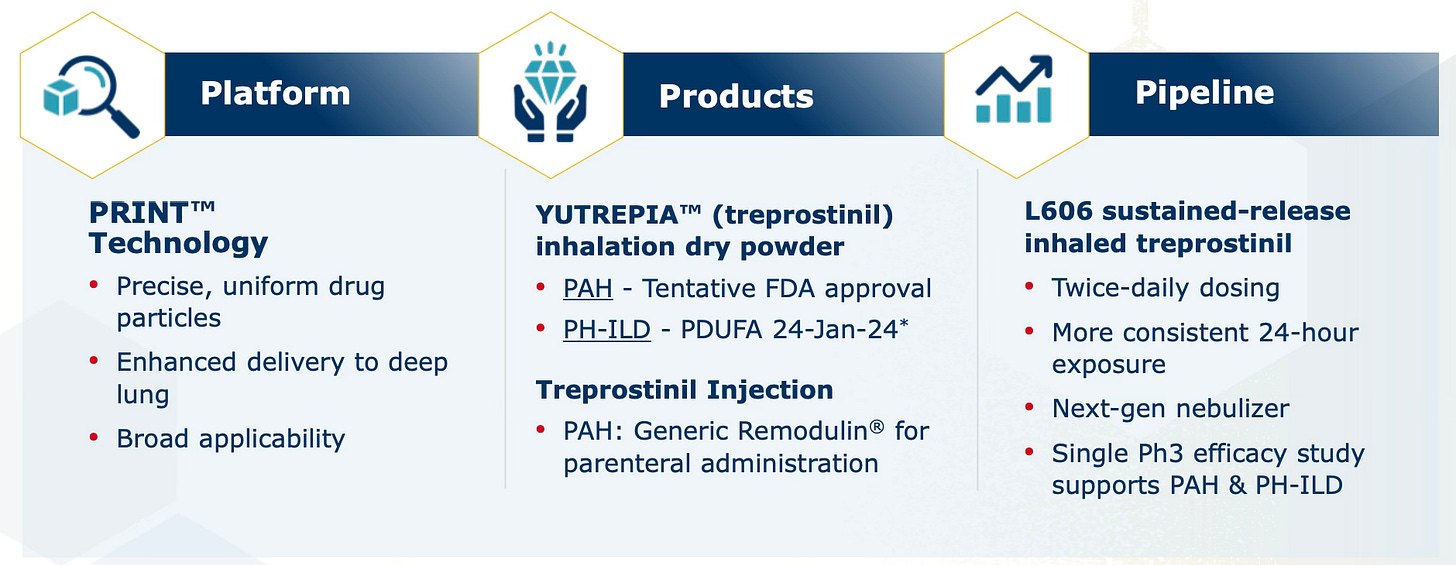
Liquidia Corporation ($LQDA) is a biopharmaceutical company focused on the development and commercialisation of innovative therapies for patients with rare and serious pulmonary diseases. The company leverages its proprietary PRINT® (Particle Replication In Non-Wetting Templates) technology to engineer drug particles with precise size, shape, and chemistry, enhancing drug delivery and performance.
Overview of Investment Opportunity
Liquidia presents a compelling investment opportunity driven by the imminent commercialisation of its lead product, Yutrepia™ (Treprostinil), a dry powder formulation designed to treat Pulmonary Arterial Hypertension (PAH) and Pulmonary Hypertension associated with Interstitial Lung Disease (PH-ILD). With recent legal victories and strong financial backing, Liquidia is poised to capture significant market share in a growing and underserved market.
Key Points of the Investment Thesis
Imminent Commercialization of Yutrepia
Yutrepia™ has received tentative FDA approval and is expected to launch soon.
The product targets PAH and PH-ILD, both of which have substantial unmet medical needs.
Yutrepia™'s dry powder formulation offers advantages over existing treatments, such as ease of use and improved patient compliance.
Strong Market Potential
The PAH market is projected to reach $8.7 billion by 2027, driven by increasing prevalence and the need for better treatment options.
PH-ILD, affecting 30,000 to 40,000 patients, is an emerging market with significant growth potential, given the high unmet need and lack of effective therapies.
Legal and Regulatory Milestones
Liquidia recently won a critical patent infringement lawsuit against United Therapeutics ($UTHR), removing significant legal hurdles.
With no current legal impediments, Yutrepia™ is well-positioned for final FDA approval and market entry.
Experienced Management Team
Liquidia's management team, led by CEO Dr. Roger Jeffs (Ex President and co-CEO at United Therapeutics), brings extensive experience in biopharmaceutical commercialisation and a proven track record of success.
Financial Health and Backing
Patient Square Capital, a prominent healthcare investor, invested $75 million in Liquidia, acquiring nearly a 10% stake at $10.4 per share.
Potential Target for acquisition
Given Liquidia's PRINT® technology, product pipeline with and exceptional leadership, it holds strong potential to become an attractive acquisition target for larger pharmaceutical companies in the future as it progresses through commercialisation and market expansion.
Analysts predict that Yutrepia™ could capture a substantial market share from Tyvaso™ (we will talk about it extensively later in the article, however just to give you a spoiler it is UTHR's current cure for PAH and therefore the main competitor for Yutrepia™), potentially splitting the market 50/50 over the next five years. If Yutrepia™ successfully penetrates the market, Liquidia could see its market cap soar to $5 billion in the next 5 years, a significant increase from its current valuation. Beyond PAH, Yutrepia™ is positioned to treat other pulmonary hypertension-related conditions, such as PH-ILD and PH-COPD.
2. Company and Market Overview
History and Background
Liquidia Corporation was founded in 2004 and is headquartered in Morrisville, North Carolina. The company was established to develop and commercialise innovative therapies using its proprietary PRINT® technology, which allows for the precise engineering of drug particles. This technology is a core asset for Liquidia, enabling the development of therapies with improved efficacy, safety, and patient compliance.
PAH Overview and Market
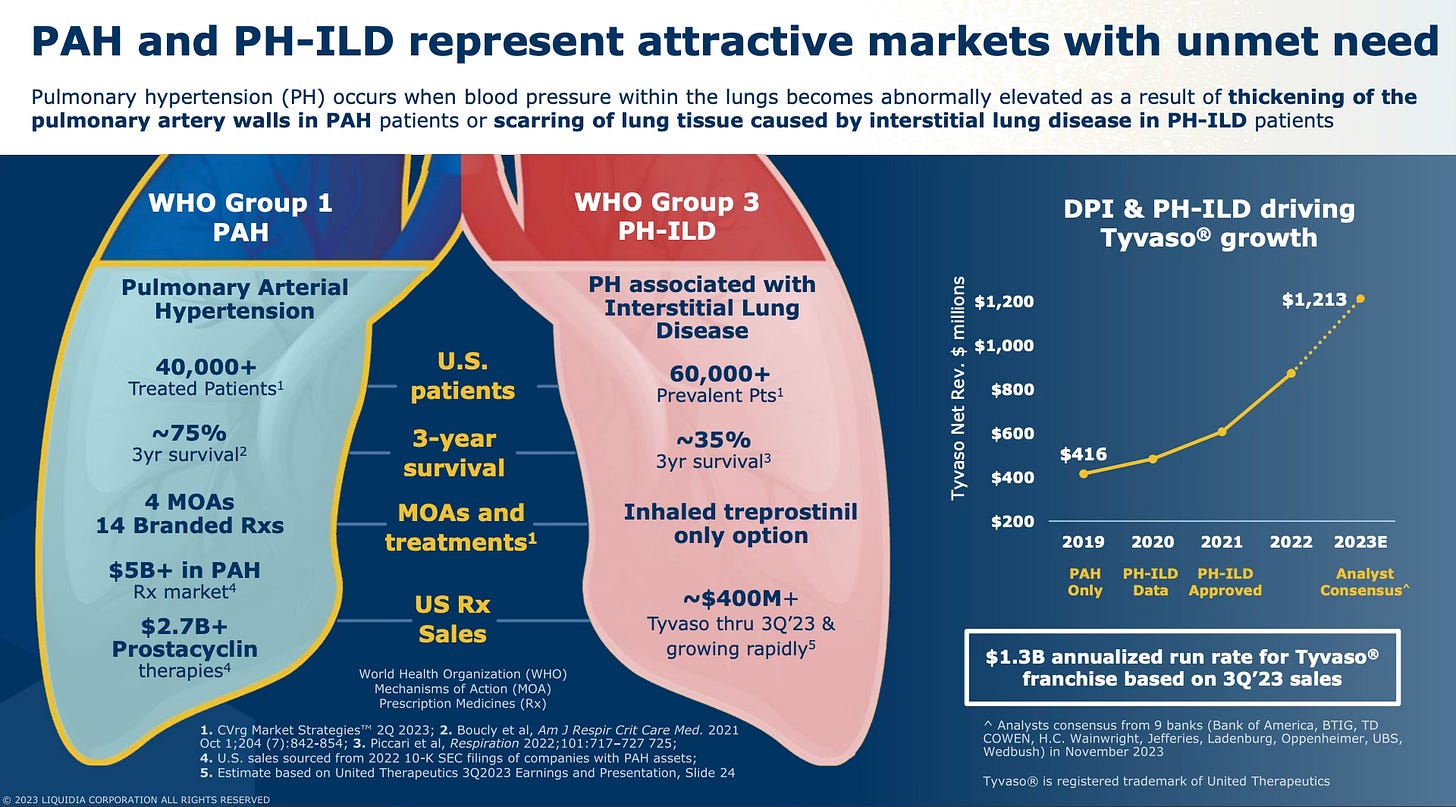
Pulmonary arterial hypertension (PAH) is a rare and progressive disease characterized by high blood pressure in the arteries of the lungs. This condition leads to the narrowing and stiffening of these arteries, which increases resistance to blood flow and forces the heart to work harder to pump blood through the lungs. Over time, this increased workload can cause the right side of the heart to weaken and eventually fail.
PAH affects individuals globally, with an estimated prevalence of half a million people. In the United States alone, approximately 50,000 patients are currently undergoing treatment for this condition.
The causes of PAH are diverse and can be categorized into several groups:
Idiopathic PAH (IPAH): PAH with no identifiable cause.
Heritable PAH: linked to genetic mutations.
Drug- and Toxin-Induced PAH: associated with exposure to certain drugs or toxins.
PAH Associated with Other Conditions: includes connective tissue diseases (e.g., scleroderma, lupus), HIV infection, portal hypertension, congenital heart diseases, and other systemic disorders.
Recent trends indicate an increase in PAH diagnoses, attributed to factors such as aging populations and lifestyle-related issues. PAH is typically classified based on the severity of symptoms into four functional classes:
Class I (circa 10% of patients): patients experience no symptoms with ordinary physical activity.
Class II (circa 35% of patients): symptoms occur with ordinary activities but not at rest.
Class III (circa 45% of patients): symptoms are present with less-than-ordinary activities, significantly limiting daily activities.
Class IV (circa 10% of patients): symptoms are present at rest, and any physical activity causes discomfort.
Current Treatment Landscape
The treatment of PAH primarily involves the use of vasodilators, which are medications that help relax and widen blood vessels, reducing the pressure in the pulmonary arteries. The mainstay of PAH treatment is Treprostinil, a synthetic analogue of Prostacyclin, which can be administered through various methods:
Oral Administration: associated with significant side effects, such as gastrointestinal issues, liver toxicity, and potential teratogenic effects.
Injectable Form: administered via continuous intravenous infusion, requiring the surgical placement of a venous catheter. This method carries risks of infection and other complications.
Inhalation Therapy: treprostinil can be inhaled using a nebulizer or dry powder inhaler (DPI). Nebulizers, which are bulky and require frequent dosing, are being increasingly replaced by more convenient DPIs.
Key Drugs in the PAH Market
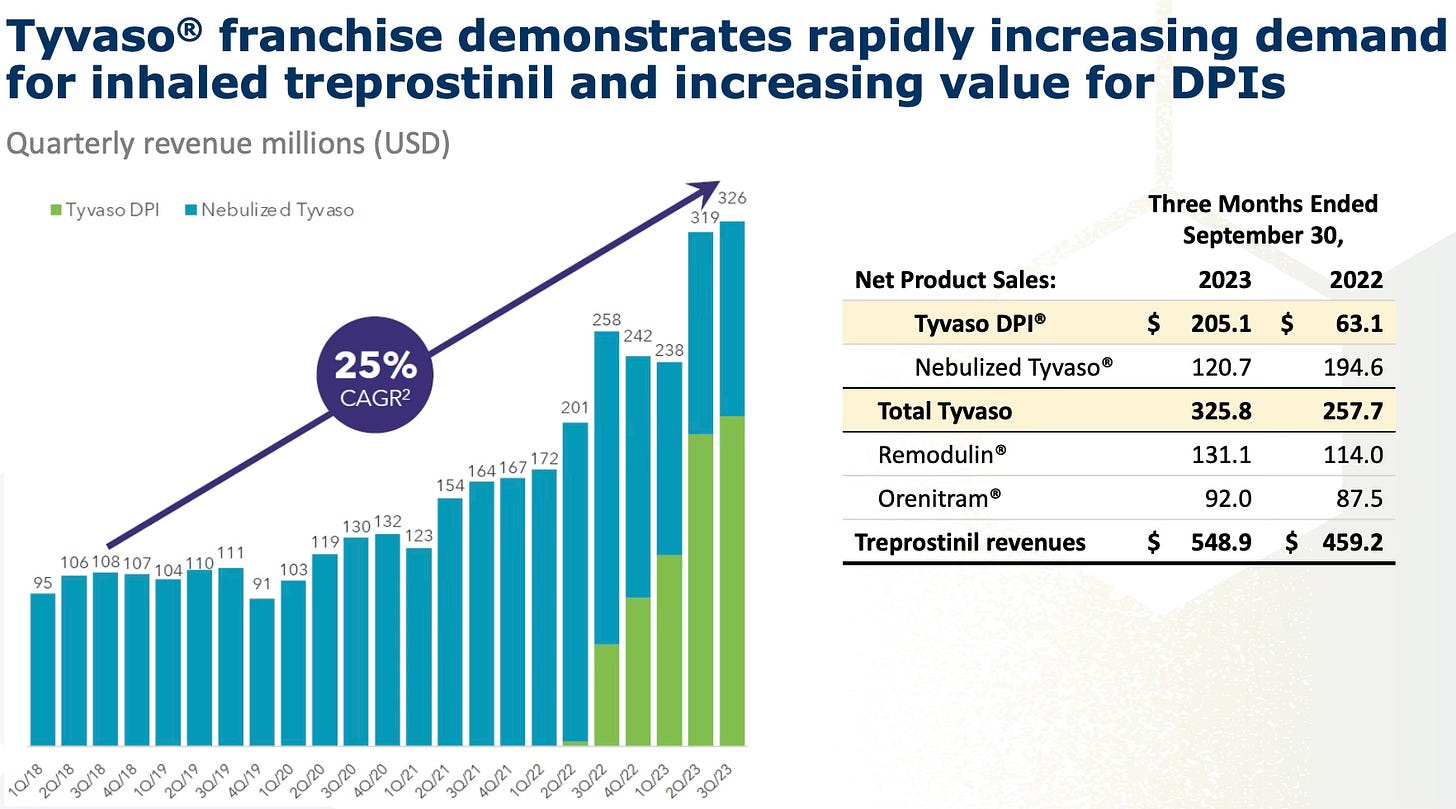
Tyvaso™ (Treprostinil): developed by United Therapeutics ($UTHR), Tyvaso™ is available in both nebulized and DPI forms. Tyvaso™ DPI, which recently received FDA approval, offers a smaller, more portable inhaler option, improving patient compliance and quality of life. In the second quarter of 2023, Tyvaso™ generated $319 million in revenues, representing 54% of UTHR's total revenues, and is the fastest-growing segment for the company.
Yutrepia™ (Treprostinil): Liquidia's Yutrepia™ is a DPI formulation that competes directly with Tyvaso™ DPI. Yutrepia has demonstrated potential advantages, including higher tolerable doses and a low-resistance inhaler, making it suitable for patients with severe breathing difficulties, such as those with PAH-ILD (pulmonary hypertension associated with interstitial lung disease).
Market Dynamics and Potential
The PAH market, valued at over $5 billion, is primarily driven by the need for effective and convenient treatment options. UTHR's Tyvaso™ and Liquidia's Yutrepia™ are key players in this market. The inhalation route, especially with DPIs, is favored due to its ease of use, fewer side effects, and better patient adherence compared to oral and injectable forms.
The market for PAH treatments is expanding beyond traditional PAH indications. Tyvaso™ has been approved also for PH-ILD (pulmonary hypertension associated with interstitial lung disease), a condition affecting more than 60,000 patients, and is in advanced trials for PH-COPD (pulmonary hypertension associated with chronic obstructive pulmonary disease), potentially addressing a broader patient population. Additionally, Tyvaso™ is being tested for idiopathic pulmonary fibrosis (IPF), which affects approximately 200,000 individuals.
The competitive landscape for PAH treatments is currently dominated by UTHR, with Liquidia poised to disrupt the market with Yutrepia™. While Tyvaso™ has established a strong presence, Yutrepia™'s entry, supported by better delivery technology and potential clinical benefits, is expected to capture significant market share. The differentiation between the two products lies in their formulation and delivery mechanisms, with Yutrepia™'s PRINT® technology offering a distinct advantage.
Management Team
Roger Jeffs, Ph.D. - Chief Executive Officer: Dr. Jeffs brings over three decades of experience in the biopharmaceutical industry. He previously had a 18 years tenure at UTHR. He joined United Therapeutics Corporation during its start-up phase in 1998 as Director of Research, Development, and Medical, and served as the company’s President and Chief Operating Officer from 2001 to 2014 and President & co-CEO from 2015-2016.
Michael Kaseta - Chief Financial Officer: Mr. Kaseta has extensive experience in corporate finance and strategic planning within the biopharmaceutical sector. Prior to Liquidia, Mr. Kaseta served as the Chief Financial Officer at Aerami Therapeutics, a biotech company focused on the development of improved therapies for the treatment of severe respiratory diseases, including pulmonary arterial hypertension (PAH). Previously, Mr. Kaseta served as the Chief Financial Officer at Aralez Pharmaceuticals Inc. ($ARLZ) and spent eleven years at Sanofi in a variety of financial roles, culminating with the Chief Financial Officer role.
Rajeev Saggar, M.D. - Chief Medical Officer: Dr. Saggar is a renowned pulmonologist with significant clinical and research experience in pulmonary hypertension. He brings more than 20 years of experience as a practicing pulmonologist and has authored more than 60 peer-reviewed publications with scientific interests. He oversees Liquidia's clinical development programs and product pipeline.
3. Recent News and Developments
Liquidia has been embroiled in a complex legal battle with United Therapeutics (UTHR) over patent infringements. This section outlines the critical legal milestones that have influenced Liquidia’s path to commercialization.
Hatch-Waxman Patent Infringement Lawsuit (June 2020): in June 2020, UTHR initiated a Hatch-Waxman infringement claim against Liquidia, targeting three of its Tyvaso™ patents (‘066, ‘901, and ‘793). This lawsuit triggered a 30-month stay on the FDA's final approval of Yutrepia™, putting Liquidia’s commercialization efforts on hold.
Tentative FDA Approval (November 2021): despite the ongoing litigation, Yutrepia received tentative FDA approval in November 2021. This approval was contingent upon the resolution of the patent disputes, but it marked a significant step forward for Liquidia, validating the potential of its inhaled Treprostinil product.
Licensing Bid Rejection (October 2020): during the litigation, an undisclosed party, likely UTHR, made a licensing offer worth approximately $8 per share for Liquidia. At the time, Liquidia’s share price was $2.80, but the company declined the offer, indicating confidence in its long-term prospects.
Patent Drops and Court Rulings:
In December 2021, UTHR dropped the ‘901 patent from the lawsuit, acknowledging non-infringement by Liquidia.
In July 2022, the Patent Trial and Appeal Board (PTAB) ruled that all claims of the ‘793 patent were unpatentable.
In August 2022, a district court invalidated five of the six asserted claims of the ‘066 patent and found the remaining claim not infringed by Liquidia. However, Liquidia was found to induce infringement of the ‘793 patent, a ruling that was mitigated by the PTAB’s earlier decision.
Amendment to Add PH-ILD Indication (July 2023): on July 27, 2023, Liquidia submitted an amendment to its New Drug Application (NDA) to include the PH-ILD indication for Yutrepia™. This move aims to capitalize on the recently expired market exclusivity for Tyvaso™ in treating PH-ILD, potentially allowing Liquidia to tap into a broader patient base.
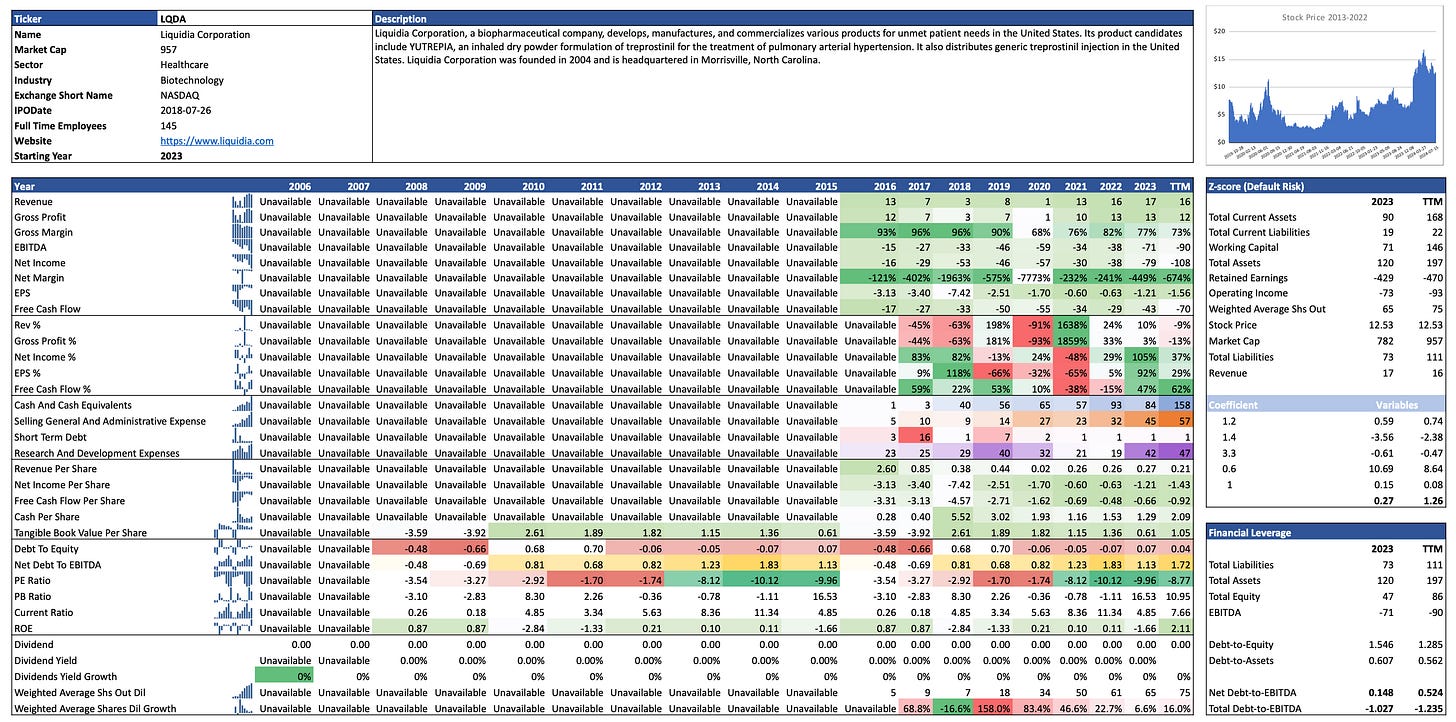
Q1 2024 Financial Results: for the first quarter of 2024, Liquidia reported revenues of $3 million, a decrease from $4.5 million in the same period of 2023. This decline was primarily due to lower sales quantities and adjustments from the previous year. However, the company increased its investment in research and development and expanded its commercial infrastructure in anticipation of Yutrepia™’s approval.
District Court Issues Favorable Ruling and Denies United Therapeutics’ Request to Block Yutrepia™ Launch (June 2024): Judge Andrews of the U.S. District of Delaware denied the motion for preliminary injunction filed by United Therapeutics in September 2023 in which it has alleged Yutrepia™ would infringe U.S. ‘327 patent, that sought to block the launch. The ruling reinforces the clear path for the U.S. Food and Drug Administration (FDA) to issue a final decision on the amended New Drug Application (NDA) for Yutrepia™.
Dr. Roger Jeffs said: “We are pleased that Judge Andrews ruled that United Therapeutics’ request for an injunction failed on critical grounds, including UTHR’s failure to show our obviousness challenge lacks substantial merit and UTHR’s failure to show that the public interest weighs in favor of an injunction. While we await a final FDA action, we will continue to intensify our commercial preparations as we work to make this important treatment option available to pulmonary arterial hypertension (PAH) and PH-ILD patients.”
Commercialization Plans: the company has onboarded a sales force of 50 representatives, equipped with extensive experience in rare diseases and pulmonary hypertension.
Is Liquidia’s recent legal triumph the David-vs-Goliath moment we’ve been waiting for, setting the stage for Yutrepia™ to conquer the market and silence the skeptics? I guess we will soon find out!
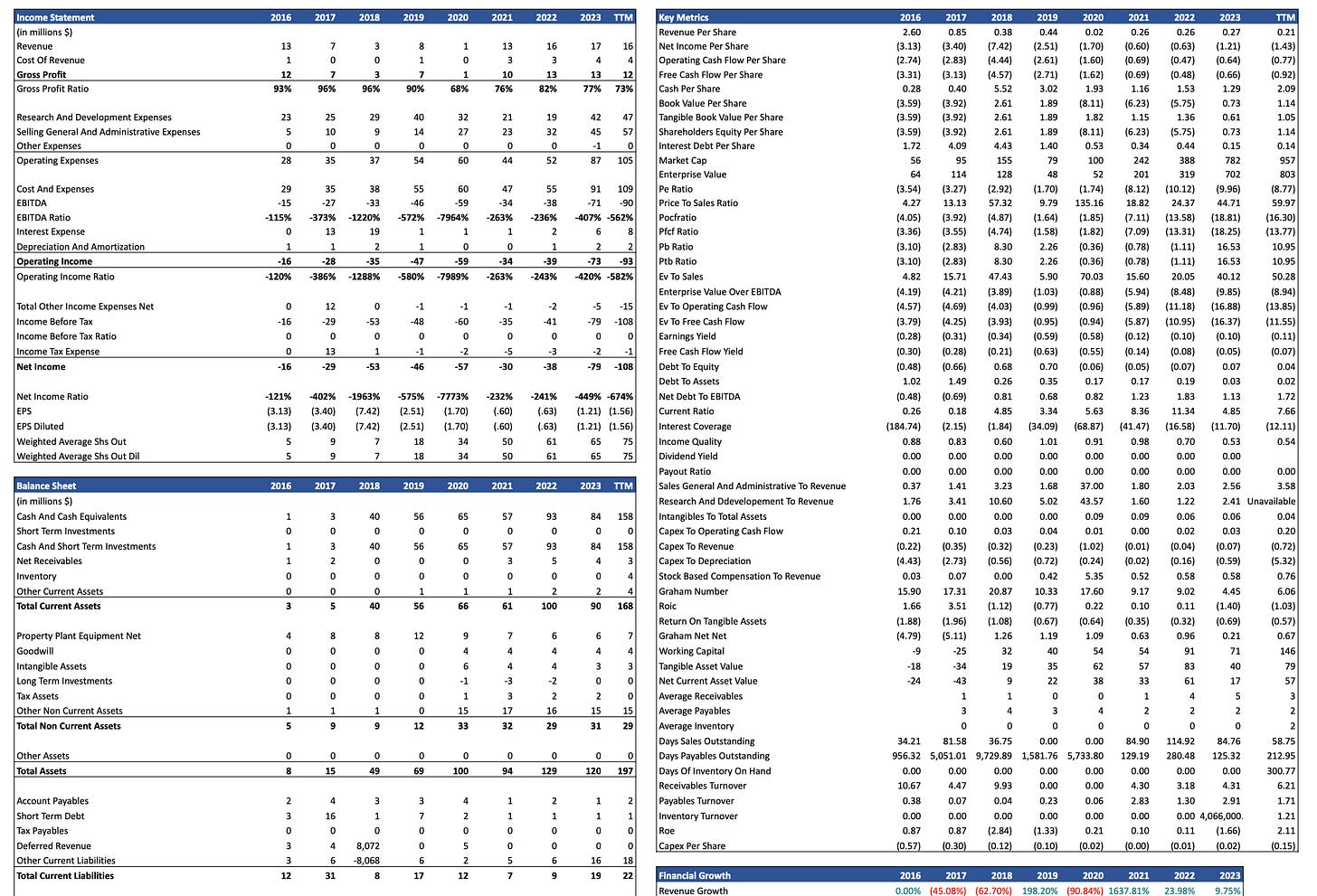
5. Detailed Financial Analysis
In this chapter, we delve into the financials of Liquidia to evaluate its intrinsic value. Let’s walk through the key components of this valuation.
Introduction to the Valuation Model
The valuation model aims to capture the fundamental worth of Liquidia by examining various aspects of its financial and operational performance. The model includes:
Historical Financial Performance: analysis of revenue, profit margins, and key financial ratios over the past years.
Financial Statements, Estimates and Key Metrics: detailed assessment of the company Balance Sheet, Income Statement and Cash Flow, including some analyst’s estimates.
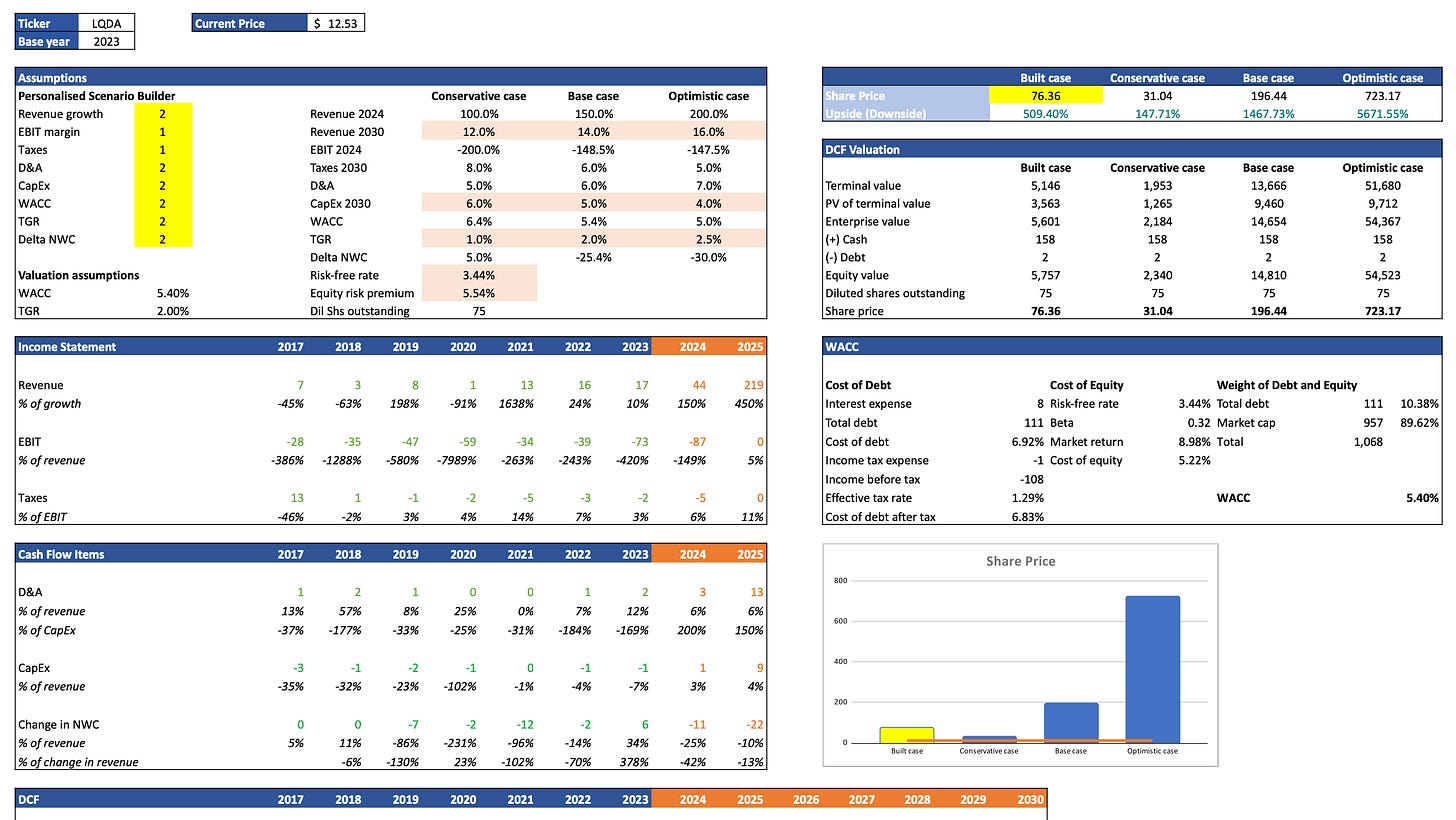
Discounted Cash Flow (DCF) Analysis: calculation of the present value of expected future cash flows using a personalised scenario builder.
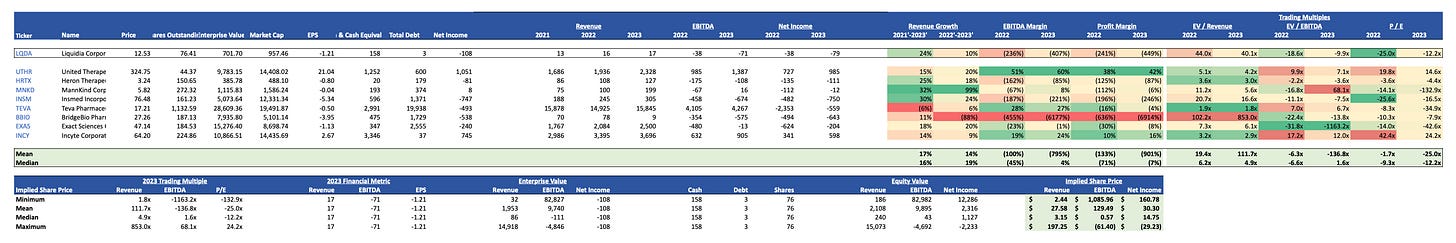
Trading Comparables: evaluation of the company's metrics relative to its industry peers. Insights into LQDA's market positioning and potential valuation.
6. Valuation Analysis - My 2 cents
LQDA will target UTHR's crown jewel for the treatment of PAH. Analysts predict that within 5 years the market for PAH cures will be split split 50% for Tyvaso™ and 50% for Yutrepia™. If we take into consideration that currently 70% of UTHR's market cap is backed by Tyvaso™, this means that LQDA could support a market cap of approximately USD 5 billion in 5 years (USD 14.52 billion x 70% x 50%). This is a 5.3x increase over the current market cap (950 million) at a share price of $77.25. This is not even an exceptional case where LQDA captures 50% of the market, as it is expected to grow and according to recent publications Yutrepia™ will also be prescribed for the treatment of PH-ILD and PH-COPD.
So how long could it take to see this appreciation? I would say at least as long as it takes to see the market launch, the rapid adoption and the first financial figures coming in.
If you made it to the end I can only thank you! I hope you have found this article useful and enjoyed it, I will try to be as consistent as possible with the publication and when there are updates I will write another article.
If you would like to support me in this work I would be very glad to receive a like and if you would like to share the post you would give me an incredible contribution to make Strategic Alpha known to as many people as possible!
See you soon
Attilio
*Disclaimer
The information provided on Strategic Alpha - a Special Situations Gazette is for informational purposes only and should not be construed as financial advice. While I may hold positions in some of the stocks discussed, I do not take responsibility for any decisions made based on the content of this site. It is crucial for readers to conduct their own due diligence and consult with a professional financial advisor before making any investment decisions.
The trademarks and registered trademarks mentioned in this article include, but are not limited to, Liquidia Corporation®, Yutrepia™, PRINT® technology, United Therapeutics®, Tyvaso™, and Tyvaso DPI™. All other trademarks and registered trademarks are the property of their respective owners.